Recce Pharmaceuticals Ltd (ASX:RCE, OTC:RECEF) has met all primary endpoints in a Phase 1/11 clinical trial assessing RECCE® 327 (R327) as a broad-spectrum anti-infective topical treatment for patients with mild skin and soft tissue diabetic foot infections (DFI).
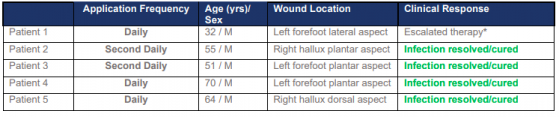
The trial showed R327 well-tolerated in all patients who were treated either daily or every second day for 14 days, with their DFIs resolved or cured.
Investors have welcomed the positive news, with Recce shares trading 10.2% higher at $0.54 in late morning ASX trading.
Summary of treated patients.
“We are pleased that the Phase I/II clinical trial has met all primary endpoints and produced efficacy data to support R327 to be used as a topical agent,” Recce chief executive officer James Graham said.
“We look forward to expanding the study by accessing a global patient population and further enhancing our portfolio of human efficacy data.”
High treatment cost
Diabetes is the leading cause of non-traumatic lower extremity amputations in the United States, with 14-24% of patients with diabetes who go on to develop a foot ulcer requiring amputation.
About 85% of diabetes-related amputations are due to foot ulceration.
Treating diabetic foot diseases in the US costs US$9-13 billion every year.
Read more on Proactive Investors AU