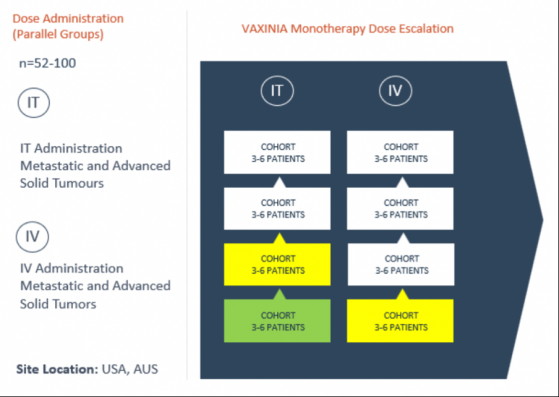
Imugene Ltd (ASX:IMU, OTC:IUGNF) has dosed the first intratumoral (IT) patient in the second cohort of its clinical trial, designed to study the novel, cancer-killing VAXINIA virus.
VAXINIA (technically known as CF33-hNIS) is at the centre of a multi-stage Phase 1 metastatic advanced solid tumours (MAST) study, which evaluates the safety of this oncolytic virus.
Back in September, Imugene dosed the first patient in its maiden intravenous (IV) cohort, setting an important arm of the monotherapy dose escalation study in motion.
So far, the study has delivered a low dose of the virus to patients in Australia and the US with metastatic or advanced solid tumours who have had at least two prior lines of standard-of-care treatment.
It’s still early days, but the City of Hope-developed oncolytic virus has been shown to shrink colon, lung, breast, ovarian and pancreatic cancer tumours in pre-clinical laboratory and animal models.
Imugene’s multi-stage Phase 1 trial is expected to run for roughly 24 months and is funded from existing budgets and resources.
Commenting on the move to IT cohort two, Imugene CEO and managing director Leslie Chong said: “The VAXINIA trial continues to progress on schedule and we’re very excited to see the results it can deliver for these patients dealing with significant tumour growth.”
Read more on Proactive Investors AU