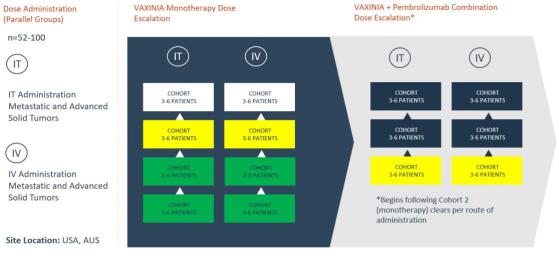
Imugene Ltd (ASX:IMU, OTC:IUGNF) has progressed its Phase 1 MAST (metastatic advanced solid tumours) study evaluating the safety of novel cancer-killing virus CF33- hNIS (VAXINIA) to the next stage, clearing cohort 2 of both the intravenous (IV) and intratumoral (IT) arms of the monotherapy trial.
The company’s success has allowed it to open cohort 1 of the combination study (with Pembrolizumab, an immune checkpoint inhibitor antibody) and cohort 3 for both arms of the monotherapy dose escalation portion of the trial.
VAXINA dose escalation outline.
The first patient was dosed in IV cohort 2 on December 5, 2022, and the trial has continued unimpeded since.
Early indications positive
“Early data arising from our patients dosed at low levels with our CF33 oncolytic virus have indicated immune activation is occurring in the tumour microenvironment, turning the tumour from ‘immunologically cold to hot’,” Imugene managing director and CEO Leslie Chong said.
“This is a perfect time to introduce an immune checkpoint inhibitor such as pembrolizumab.”
VAXINIA, the City of Hope-developed oncolytic virus, has been shown to shrink colon, lung, breast, ovarian and pancreatic cancer tumours in pre-clinical laboratory and animal models.
The multicenter Phase 1 MAST trial began by delivering a low dose of VAXINIA to patients with metastatic or advanced solid tumours who have had at least two prior lines of standard-of-care treatment.
Read more on Proactive Investors AU