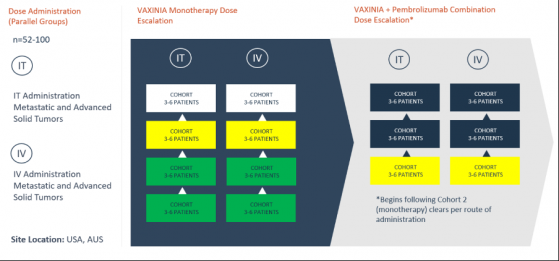
Imugene Ltd (ASX:IMU, OTC:IUGNF) has dosed the first patients in cohort three of its intravenous (IV) and intratumoral (IT) monotherapy dose-escalation trial for novel cancer-killing virus VAXINIA (also known as CF33-hNIS).
As diagrammed below, Imugene’s study continues on schedule — the first cohort in the company’s VAXINIA and Pembrolizumab combination study has also been dosed.
Imugene managing director and CEO Leslie Chong said: “Still less than 12 months since the very first patients were dosed, we’re now well advanced on amassing the critical data we require to publish on the outcomes of this study and we remain very positive on the potential benefit to patients.”
Study background
IMU’s phase one metastatic advanced solid tumours (MAST) study began in May last year by delivering a low dose of VAXINIA to patients with metastatic or advanced solid tumours who have had at least two prior lines of standard-of-care treatment.
The City of Hope-developed oncolytic virus has been shown to shrink colon, lung, breast, ovarian and pancreatic cancer tumours in preclinical laboratory and animal models.
Overall, the study aims to recruit up to 100 patients across roughly 10 trial sites in the United States and Australia.
The trial is expected to run for 24 months, funded from existing budgets and resources.
Read more on Proactive Investors AU