Immuron Ltd (NASDAQ:IMRN, ASX:IMC) welcomes an uptick in travel and holidaymaking across the globe that has generated strong March quarter sales of its Travelan® and Protectyn® gut health products.
Reporting on its unaudited financials this morning, Immuron revealed sales in Australia tripled the 1H FY23 result, while Travelan® sales in North America reached double what was generated in the last six months.
The company will also be hosting a business update webinar today, April 19, at 11:00 am AEST. Registrations can be made here.
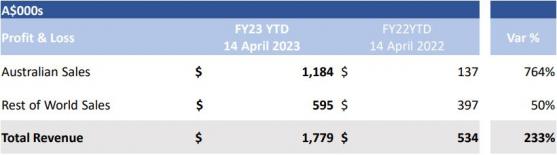
On a global scale, Immuron’s year-to-date sales are up 233% in FY23, reaching A$1.779 million compared to the A$534,000 total reported at April 14 last year.
Moving ahead, the company is focused on growing commercial product sales, expanding its portfolio and developing its robust clinical pipeline.
Australian sales growth
Immuron’s Australian product sales reached A$572,000 in the March quarter — more than triple the metrics reported in FY23’s first half.
IMC attributes the Australian sales uptick to a rebound in overseas travel. According to the Australian Bureau of Statistics, short-term departures in January 2023 reached 80% of 2019 levels, reflecting a return to pre-pandemic holidaymaking.
In the wake of heightened demand, Immuron continues to restock wholesalers in line with increasing pharmacy sales.
Travelan picks up the pace in NA
Over in North America, Immuron’s Travelan product netted just shy of A$300,000 during the quarter, doubling the revenue generated in the previous six months.
Travelan brought in A$595,000 over the last nine months, up 70% on the A$350,000 generated in the same period during FY22.
Ultimately, roaring trade across the US’ Passport Health travel clinics and growing distributor sales sparked healthy sales growth across the Travelan brand.
While not yet at its pre-pandemic peak, Immuron expects sales in the US to continue to grow as it replenishes and builds inventory.
Commercial and clinical upside
Immuron is responsible for Travelan, an over-the-counter immune supplement that can help patients curb traveller’s sickness and diarrhoea.
It also makes Protectyn, a targeted antibody treatment that works by removing harmful bacteria and LPS toxins in the gut.
Travelan is available in major pharmaceutical markets like Australia, the US and Canada, while Protectyn is gearing up for a Canadian release after launching on home soil.
In a recent interview with Proactive, Immuron CEO Steven Lydeamore said his focus was on expanding into key markets.
“Instead of selling Imodium or Gastro-Stop, which is a product that people take after they get traveller's diarrhoea, wouldn’t it be better to sell a higher price product that benefits the patient?” Lydeamore asks.
“When you sit back and look at the target population — that’s people travelling to high-risk regions — there’s no reason why we couldn’t grow that product itself to more than $50 million."
Immuron’s growth is not restricted to its commercial pipeline: Lydeamore also has plans to advance the company’s clinical prospects.
The CEO recently engaged one of the US’ leading consulting companies to research Immuron’s market potential. A mix of desktop and primary research revealed the revenue base case (in the US alone) for each of Immuron’s products amounted to US$100 million per annum.
“We’re committed to the market opportunities there while we advance our clinical programs,” Lydeamore reveals.
Read more on Proactive Investors AU