Emyria Ltd (ASX:EMD) has signed a deal with Aspen Pharmacare (JSE:APN) Australia for the licensing and commercialisation of EMD-RX5.
The agreement supports the registration and commercialisation of EMD-RX5 as an over-the-counter (OTC) treatment in Australia.
Leading pharma company
Aspen Australia is one of Australia’s leading pharmaceutical companies, and a subsidiary of Aspen Pharmacare Holdings, a multinational pharmaceutical company with a market cap in the order of A$6.7 billion.
Emyria will receive a royalty of up to 10% based on annual net sales, with aggregate milestone payments of $400,000 for satisfying agreed milestones.
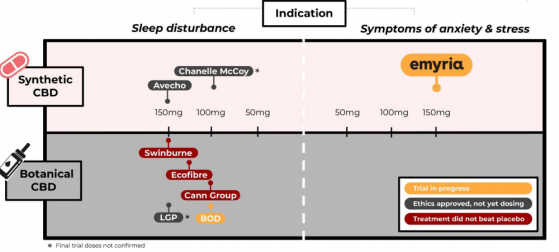
EMD-RX5 is Emyria’s first, ultra-pure CBD capsule targeting Therapeutic Goods Administration (TGA) registration as an over-the-counter (OTC) treatment for the symptoms of mild stress and anxiety in patients with a chronic health condition.
EMD-RX5 is the subject of Phase 3 clinical trials and Emyria hopes it will be the first CBD product registered as an OTC treatment in Australia.
It is a solid capsule-form medication, a preferred dosage form for patients and prescribers.
Large potential market
The treatment has a large potential patient market.
Mild anxiety and stress affect up to 15% of the adult population, with a higher prevalence in patients with chronic disease, such as chronic pain, which affects around 2.8 million adults in Australia.
“Emyria expects to become one of the first companies to register a CBD product for the rapidly emerging Australian OTC market, thanks to our innovative product and registration strategy,” managing director Dr Michael Winlo said.
“We are excited to collaborate with Aspen, one of Australia's largest and most successful pharmaceutical companies with an impressive track record in sales, marketing, manufacturing and distribution.
“Aspen's extensive sales force is unique in that it can reach both pharmacies and prescribing doctors, which will be instrumental in ensuring that Emyria's product has strong brand awareness and gains widespread adoption after registration.”
About the deal
Aspen Australia is a subsidiary of Aspen Holdings, which has a portfolio of more than 140 brands of both prescription and OTC medicines covering a wide range of therapeutic categories.
The company has significant sales and marketing capabilities, with a team of around 80 sales and marketing heads to support the successful commercialisation of EMD-RX5.
Aspen Australia will provide funding and resources to support the development and regulatory approvals for EMD-RX5 in Australia. Accordingly, it will have the exclusive right to market, sell and distribute EMD-RX5 in Australia.
Aspen Australia has full manufacturing capabilities at its site in Dandenong, Victoria, and will retain the rights to evaluate the opportunity to manufacture EMD-RX5 locally.
Read more on Proactive Investors AU